
But they're not as reactive as the alkali metals. Each of these elements has two electrons in its outermost energy level, which makes the alkaline earths reactive enough that they're rarely found alone in nature. Hydrogen, with its single electron, also lives in Group 1, but the gas is considered a nonmetal.Īlkaline-earth metals: The alkaline-earth metals make up Group 2 of the periodic table, from beryllium (Be) through radium (Ra). They are also extremely reactive and will burst into flame or even explode on contact with water, so chemists store them in oils or inert gases. Shiny and soft enough to cut with a knife, these metals start with lithium (Li) and end with francium (Fr). Column P consists of the colors in the PubChem database managed by the United States National Institute of Health.Īll colors are approximate and may depend on the display hardware and viewing conditions.The periodic table of elements is arranged into several broad groups (Image credit: Future) Groups of the Periodic tableĪlkali metals: The alkali metals make up most of Group 1, the table's first column.Column R is the scheme used by Rasmol when two colors are shown, the second one is valid for versions 2.7.3 and later.Column J is the color scheme used by the molecular visualizer Jmol.Column C is the original assignment by Corey and Pauling.The following table shows colors assigned to each element by some popular software products. carbon black, hydrogen white, chlorine green, 'fiery' oxygen red, nitrogen blue)." Modern variants Example of Jmol coloring (At the time, croquet was the most popular sport in England, so the balls were plentiful.) "On the Combining Power of Atoms", Chemical News, 12 (1865, 176–9, 189, states that "Hofmann, at a lecture given at the Royal Institution in April 1865 made use of croquet balls of different colours to represent various kinds of atoms (e.g. :max_bytes(150000):strip_icc()/PeriodicTableallcolor-58b5d9293df78cdcd8d043b6.jpg)
In 1865, August Wilhelm von Hofmann, in a talk at the Royal Institution in London, was using models made from croquet balls to illustrate valence, so he used the coloured balls available to him. It is likely that the CPK colours were inspired by models in the nineteenth century. Perhaps red for oxygen is inspired by the fact that oxygen is normally required for combustion or that the oxygen-bearing chemical in blood, hemoglobin, is bright red, and the blue for nitrogen by the fact that nitrogen is the main component of Earth's atmosphere, which appears to human eyes as being colored sky blue. For some colors, such as those of oxygen and nitrogen, the inspiration is less clear. For example, hydrogen is a colorless gas, carbon as charcoal, graphite or coke is black, sulfur powder is yellow, chlorine is a greenish gas, bromine is a dark red liquid, iodine in ether is violet, amorphous phosphorus is red, rust is dark orange-red, etc. 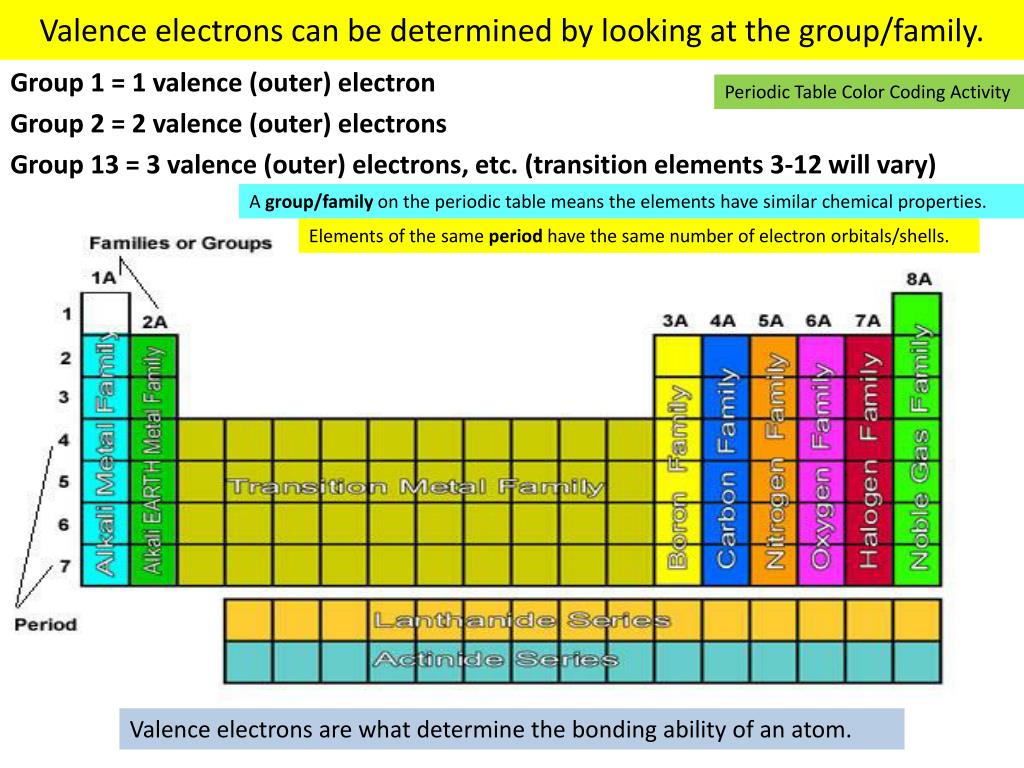
Several of the CPK colors refer mnemonically to colors of the pure elements or notable compound. ( October 2022) ( Learn how and when to remove this template message)Ī box of ball-and-stick model pieces colored to represent several of the common elements.Īlkaline earth metals ( Be, Mg, Ca, Sr, Ba, Ra) Statements consisting only of original research should be removed. Please improve it by verifying the claims made and adding inline citations. This section possibly contains original research.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
